 |
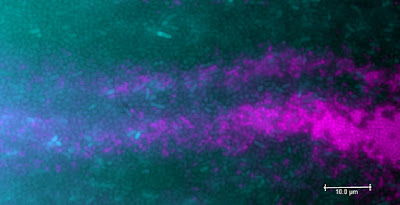
| Cooperative bacterial strains colonizing a surface |
Bacteria colonize surfaces in all environments. That could be the surfaces of soil aggregates, of rocks in a stream bed, of plant leaves, of animal skin, or that could be the surface of your showerhead... On such surfaces microbes establish complex communities ('biofilms') that can contain many different interacting species. These various species are usually not randomly distributed in the biofilm, but rather organized depending on their environmental preferences (for example some like well-aerated areas, others not so much...) and on the type of interactions that they have with each other. This can result in complex patterns of organization that manifest at the microscopic scale and up to the millimeter scale. Such patterns are not trivial, as they can sustain microbial activity and functions that would not be possible in a well-mixed environment, which has importance for biotechnology applications as well.
In a new study published this month, we examined the role of cooperation in shaping spatial patterns of bacterial organization on wet surfaces. The paper is available online and is entitled '
Cooperation in carbon source degradation shapes spatial self-organization of microbial consortia on hydrated surfaces'. Our idea was that a feeding dependency between two partners would directly control their distribution in space, hence imposing a specific pattern. We used a simple model system made of two bacterial strains that could grow using the chemical compound toluene (a hydrocarbon), but only when they were working together as a 'team' (a bacterial consortium in the jargon).
